Welcome to the CoreX Regulatory Consulting GmbH in Freiburg
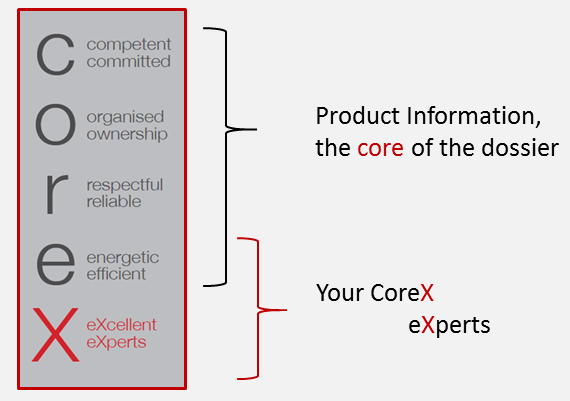
CoreX is a specialised regulatory service provider in the Health Care environment.
We support companies with regulatory services in the context of Marketing Authorisation Applications or Post-Approval Applications at European Health Authorities. The focus of our services is on the set up and management of labelling documents for medicinal products in English and all EU/EEA languages required by European Regulations.
CoreX relies on the expertise of the company’s core team, with in total more than 50 years of relevant experience, gained as consultants, in industry and at a Health Authority.